- Home
- About
- Contact
- Speed reader pl 2-0 chomikuj
- Desktop toys machine gun
- Covid vaccine insomnia
- Parallels desktop 14 dmg
- Favelas vox youtube
- Lattice energy of nacl
- Crucial conversations summary
- Usb c to usb adapter
- In the hall of the mountain king virtual piano
- Final fantasy iv
- Bombyx mori life cycle ppt
- Vlc streamer full version ipa
- Empire total war ai mod
- Vessel volume calculator
- Retroarch core
The lattice enthalpy of an ionic compound is the enthalpy change which occurs when one mole of an ionic compound dissociates into its ions in gaseous state.What is lattice energy explain how can you determine lattice energy of NaCl by using Born-Haber cycle? Sodium fluoride (NaF) shows highest lattice energy among these compounds since Na+ features ions of the same charge, the lattice energy increases as the size of the ions increases. Example 2: The lattice energy of AgBr is 895 KJ mol-Īnswer: 1.Example 1: Compute the Lattice energy of NaCl by using Born-Lande equation.Lattice Energy Formula per mole is symbolized as.The value of lattice energy of, -785 kJ/mole and -923 Kj/mole respectively. Consequently, NaCl and NaF molecules are LiF has highest lattice energy. Is Now, lattice energy directly proportional to the multiple of ionic charge and inversely proportional to the radius of cation and anion. Does NaCl have the highest lattice energy? Because the Li+ ion is smaller than the Na+ ion, the Coulombic attractions between ions in LiCl are stronger than in NaCl. The size of alkali metals increases from Li to Cs. Lattice energy depends on the size of ion and it decreases with increase in size. Which has the highest lattice energy LiCl NaCl KCl Rbcl? ∆H4 = Electron affinity of Cl(s) = – 349 kJ mol-1.∆H3 = dissociation energy of Cl2(s) = 244 kJ mol-1.∆H2 = ionisation energy of Na(s) = 495 kJ mol-1.∆H1 = heat of sublimation of Na(s) = 107 kJ mol-1.∆Hf = heat of formation of sodium chloride = 413 kJ mol-1.How do you find the lattice energy of NaCl? The lattice energy of NaCl, for example, is 787.3 kJ/mol, which is only slightly less than the energy given off when natural gas burns. The lattice energies of ionic compounds are relatively large. U = (–411.3) – (108.7 + 495.Are you looking for the answer to the question: What Is the Lattice Energy of Sodium Chloride? We have gathered for you the most accurate and comprehensive information that will fully answer the question: What Is the Lattice Energy of Sodium Chloride? ΔH 3 = dissociation energy of Cl 2(S) = 244 kJ mol –1 ΔH 2 = ionisation energy of Na(S) = 495.0 kJ mol –1 ΔH 1 = heat of sublimation of Na(S) = 108.7 kJ mol –1 ΔH f = heat of formation of sodium chloride = – 411.3 kJ mol –1 Let us calculate the lattice energy of sodium chloride using Born-Haber cycle The sum of the enthalpy changes of these steps is equal to the enthalpy change for the overall reaction from which the lattice enthalpy of NaCl is calculated. Also, the formation of NaCl can be considered in 5 steps. Since the reaction is carried out with reactants in elemental forms and products in their standard states, at 1 bar, the overall enthalpy change of the reaction is also the enthalpy of formation for NaCl. Let us use the Born - Haber cycle for determining the lattice enthalpy of NaCl as follows: ˆ†H f - enthalpy change for the formation of solid MX directly form elements According to Hess's law of heat summation U - the lattice enthalpy for the formation of solid MX

ˆ†H 3- Ionisation energy for M(g) to M +(g) ∆H 4 - electron affinity for the conversion of X(g) to X -(g) ΔH 2- enthalpy change for the dissociation of ½ X 2 (g) to X(g) ˆ†H 1 - enthalpy change for the sublimation M(s) to M(g) For example consider the formation of a simple ionic solid such as an alkali metal halide MX, the following steps are considered. The Born–Haber cycle applies Hess's law to calculate the lattice enthalpy. The cycle is concerned with the formation of an ionic compound from the reaction of a metal with a halogen or other non-metallic element such as oxygen.īorn–Haber cycle is primarily used in calculating lattice energy, which cannot be measured directly.

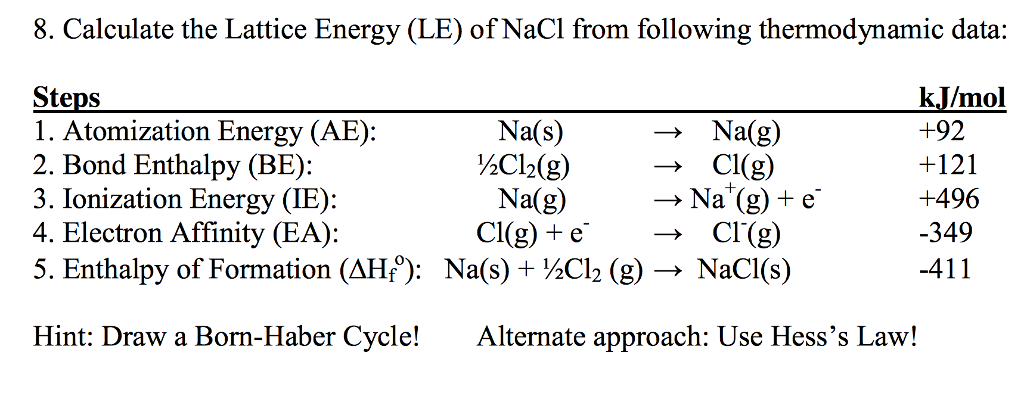
It was named after two German scientists Max Born and Fritz Haber who developed this cycle.

The Born–Haber cycle is an approach to analyse reaction energies.
